Pfizer COVID-19 Vaccine Clinical Trial Whistleblower's Documents Released
Why can't media outlets like MedPage Today do some reporting on vaccine clinical trials instead of resorting to silly "debunking" articles?
A few weeks back The BMJ published my investigation of documents provided by whistleblower Brook Jackson regarding data integrity issues in Pfizer’s COVID-19 vaccine clinical trial, “Covid-19: Researcher blows the whistle on data integrity issues in Pfizer’s vaccine trial.” This investigation has been picked up by over 91 media outlets and has an Altmetric score of over 45,000, making it the most viewed article published in any science journal for 2021. (At least, according to Altmetric.)
Besides The BMJ, Jackson has been sending these documents to multiple reporters around the world and she will soon be appearing in various investigative TV programs describing her experience at Ventavia, a research contractor hired by Pfizer for their pivotal COVID-19 vaccine trial. For example, see this short but brilliant investigative documentary on Pfizergate that just appeared on Italian TV.
About a week after The BMJ published the story on Ventavia and Pfizer’s clinical trial, The BMJ updated readers about what we know thus far.
In September, Brook Jackson contacted The BMJ about problems she had encountered while employed at Ventavia Research Group. The BMJ commissioned freelance investigative reporter Paul Thacker to write up the story. The resulting article was subject to The BMJ’s usual high level editorial oversight and review. After publication, The BMJ wrote to Ventavia, Pfizer and the U.S. Food and Drug Administration (FDA) to better clarify the scope and implications of the problems identified at Ventavia, as well as what corrective measures were taken.
To date, Ventavia has not responded to The BMJ’s repeated requests for information. However, in statements to other media outlets, Ventavia has claimed that its former employee Jackson did not work on Pfizer’s covid-19 vaccine clinical trial. This claim is not true. Jackson has contacted media organisations that have published these false allegations, sharing documentation that shows her work on the trial, and asking for corrections. At the time of writing, MedPage Today has updated its article.
During the course of this investigation, Jackson has provided The BMJ with dozens of documents, photos, audio recordings, and emails. These include evidence of her involvement in the clinical trial, and communications in which senior Ventavia employees discuss concerns that the FDA might show up at their clinical sites. These records were reviewed by The BMJ’s editors, and the story was externally peer reviewed before publication.
Because many other publications plan to publish the documents Jackson gave to me and The BMJ, and to counter false claims by Ventavia that she did not work on Pfizer’s clinical trial, I am publishing some of those documents here. Unfortunately, the media outlet MedPage Today has republished Ventavia’s defamatory statement.
New Documents
Several documents show that Jackson worked on Pfizer’s clinical trial. One is an email with Jackson thanking Pfizer’s Dr. Arturo Alfaro for giving her access to the COVID-19 clinical trial software. In a second email, Ventavia’s Mercedes Livingston invites Jackson and other staff to a “clean up call” on Pfizer’s clinical trial.


Jackson is also listed as one of the many participants on the clinical trial’s delegation log for Pfizer’s vaccine. Why Ventavia would choose to lie about Jackson’s involvement in Pfizer’s clinical trial is baffling. A couple weeks ago, Jackson’s lawyer sent Ventavia a Cease and Desist letter demanding that Ventavia retract their defamatory comment and publish a retraction.
What is most troubling about Jackson’s ordeal with Ventavia is that the Food and Drug Administration (FDA) did not investigate after Jackson’s filed a complaint about the company—this, despite fears inside Ventavia that the FDA would show up.
A week before Jackson filed her complaint, she was included on several emails where Ventavia staff discussed the chaos within their Pfizer clinical trial and “sloppy documentation” by staff moving too quickly.
Jackson was then looped in on several emails where Ventavia discussed how some staff were falsifying data. The issue of falsifying data was also in a checklist of action items for Ventavia staff.
Ventavia later sent an email to all employees expressing how afraid they were of an FDA inspection.
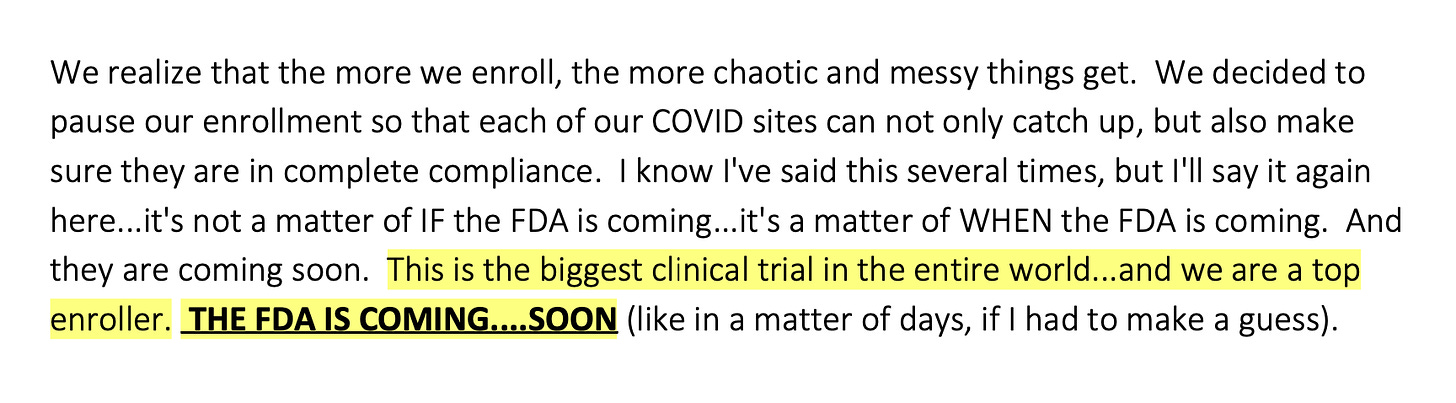
According to phone texts among Ventavia staff provided by Jackson, after Ventavia staff considered slowing down the clinical trial to manage the chaos, they then discussed how they would explain this to Pfizer. That same day, Ventavia created a form to warn staff about unblinding in Pfizer’s vaccine clinical trial.
Shortly before Jackson filed her complaint with the FDA, Ventavia sent an email to Jackson and other leadership working on the Pfizer COVID-19 vaccine clinical trial discussing problems and actions that needed to be addressed.
That next day, Jackson sent Ventavia officials an email detailing the problems she was finding in Pfizer’s clinical trial. “I might be in a little bit of shock,” Jackson ended the email.
Jackson later filed a complaint with the FDA and received a generic response acknowledging that they had received her complaint. Ventavia fired her that same day. The FDA never bothered to inspect Ventavia.
Media outlet republishes Ventavia’s defamatory comments
After The BMJ published the investigation, MedPage Today ran a pharma-friendly counter story that tried to debunk the investigation. Unfortunately for the public, debunking stories have become a popular format for journalists in recent years as they are quick to report and simply require calling upon a few experts to skim a story and offer an opinion about it.
Because MedPage Today’s debunking story carried defamatory statements that Jackson had not worked on the Pfizer clinical trial, Jackson contacted reporter Cheryl Clark and editor Kristina Fiore. Despite multiple emails from Jackson and a long, extensive interview with Clark that Jackson recorded, Fiore and Clark have failed to remove the defamatory comment. As Clark and Jackson discussed the matter in one phone conversation, Clark seemed more interested in probing Jackson’s background and credibility than understanding if anything had gone wrong at Ventavia.
“My point for you and the piece you published is that it’s defamatory,” Jackson told Clark. “And I’ve given you documentation to show that it’s inaccurate; it’s false. I would like you to take it down, Cheryl.”
Clark then started arguing and persisted in speculating that maybe Jackson had never been involved with Pfizer’s clinical trial after Ventavia hired her. “Someone could say, ‘She never got involved in that trial,’” Clark said.
In fact, Jackson sent Clark and others at MedPage Today documentation showing that she worked on the Pfizer clinical trial. Jackson says she has more documentation that details her time at Ventavia and is considering next steps—which may include having her lawyer contact MedPage Today.
For those interested in a more serious look at The BMJ’s investigation of problems at Ventavia and Pfizer’s COVID-19 vaccine clinical trial, watch this video by comedian Russell Brand that has gotten over 2.4 million views.
Seriously, a comedian did a better job than some media outlets.









Thank you for reporting on the corruption which should be addressed by the FDA.
With everything out there and easily discoverable, there should be no doubt this whole ' Shit Storm' should be shut down.